Custom Healthcare Software Development Buyer’s Guide
SeeSaw Labs//14 Min Read
A practical buyer’s guide to custom healthcare software development services—covering scope, compliance, cost, and timelines for healthcare-ready products.

If you have ever watched a healthcare software project stall, you know the moment it happens. The team has a good idea, the timeline looks reasonable, and then someone asks, “Are we dealing with PHI?” or “Does this touch an EHR?” Suddenly, everything changes.
That is why buying custom healthcare software development services is not like buying a typical business app. The stakes are higher, the workflows are messier, and compliance is not a box you check at the end.
This guide is built to help you make smart decisions early, so your scope is clear, your compliance plan is realistic, your budget is defendable, and your timeline has fewer surprises.
Who This Guide Is For
This is for you if you are any of the following:
- A healthcare operator or innovation lead buying software to improve clinical or operational workflows
- A product leader at a digital health startup planning an MVP
- A CTO, CIO, or engineering manager modernizing legacy healthcare systems
- A founder trying to avoid compliance and integration surprises
If your project involves patient data, clinical decision-making, medical devices, billing, EHR integrations, or patient-facing experiences, you are in the right place.

Scope: Define The Product Before You Price It
The fastest way to waste budget is to skip scope clarity and jump straight to estimates. In healthcare, scope is not just features; it is workflows, data boundaries, integrations, and real-world constraints.
Clarify Users, Workflows, And Outcomes
Start with three simple truths:
- Who uses it? Patients, clinicians, care coordinators, billers, admins, payers, or all of the above
- What is the job to be done? Reduce charting time, improve adherence, shorten claims cycles, close care gaps
- How will you measure success? Fewer call center tickets, faster prior auth, improved no-show rates, lower denial rates
A helpful technique is to write 5 to 10 “day in the life” workflows and identify where software can remove friction. This is where healthcare projects win or lose, because you are designing around reality, not assumptions.
If interoperability is part of your scope, you can consult the SeeSaw Labs interoperability guide.
Identify Data Boundaries (PHI, PII, Clinical Data)
Your software requirements change dramatically based on what data you create, receive, maintain, or transmit.
Ask:
- Will the system store or transmit ePHI?
- Are you ingesting data from an EHR, lab, payer, or device?
- Do you need auditability, clinical traceability, or evidence for regulators?
Even “simple” products become complex when they touch identity, clinical records, or regulated workflows.
Decide Build Vs Buy Vs Hybrid
In healthcare, “buy” can be tempting, but not always realistic. A simple rule:
- Buy when the workflow is standard and a trusted product fits 80 percent of your needs
- Build when your workflow is a differentiator, or integration requirements make off-the-shelf tools bend until they break
- Go hybrid when you can leverage proven components (video, messaging, payments, identity) but still need a custom core
Scope Checklist Table
| Scope Area | Questions To Answer | Output You Need |
|---|---|---|
| Users And Roles | Who logs in, and what do they do? | Role list, permissions matrix |
| Workflows | What happens step-by-step today? | Workflow maps, pain points |
| Data And PHI | What data is created, stored, and shared? | Data inventory, PHI boundaries |
| Integrations | EHR, payer, labs, devices, SSO? | Integration list, priority order |
| Reporting | What decisions depend on the data? | KPI list, reporting mockups |
| Non-Functional Needs | Uptime, performance, audit logs, SLAs | NFR document |
| MVP Definition | What must launch first? | MVP scope, phased roadmap |
If you want a reference for timelines on MVPs, SeeSaw Labs notes that most MVPs launch within 12 to 16 weeks, depending on scope.
Compliance: What “Healthcare-Ready” Really Means
Compliance is not one thing. It is a set of obligations that depend on your users, geography, product behavior, and data.
The goal is not to memorize regulations. The goal is to identify what applies, then build a delivery plan that proves it.
HIPAA Security Rule Basics (Administrative, Physical, Technical)
If your product handles electronic protected health information in the US, the HIPAA Security Rule is foundational. It requires regulated entities to implement reasonable and appropriate administrative, physical, and technical safeguards to protect ePHI.
In practical software terms, buyers typically expect:
- Technical safeguards like access controls, audit controls, integrity controls, and transmission security
- Administrative safeguards like risk analysis, policies, and workforce procedures
- Physical safeguards that cover devices and environments, even if you are cloud-hosted
A good partner will not just say “HIPAA compliant.” They will describe how safeguards show up in architecture, testing, and operating procedures.
Breach Notification Readiness
Healthcare buyers also care about what happens when something goes wrong. The HIPAA Breach Notification Rule requires covered entities and their business associates to notify the Department of Health and Human Services (HHS) following a breach of unsecured PHI.
That means you should plan for:
- Security monitoring and incident response workflows
- Logging that supports investigation and reporting
- Clear ownership across vendors and internal teams
Interoperability And Information Blocking (Cures Act)
If you are building patient access features, data sharing, or APIs, interoperability requirements may shape your scope.
ONC’s Cures Act Final Rule is designed to support the secure access, exchange, and use of electronic health information and to address information-blocking practices.
Buyer implication: You may need to support standardized APIs, data export, and patient access patterns earlier than you think, especially if you plug into certified health IT ecosystems.
Standards: HL7 FHIR And Why Buyers Ask For It
FHIR (Fast Healthcare Interoperability Resources) is a widely used standard for exchanging healthcare information electronically.
If someone on your buying committee says, “We need FHIR,” they usually mean one of these:
- They need to integrate with modern EHR APIs
- They want data models that map to real clinical concepts
- They have downstream analytics or care coordination needs that depend on consistent data formats
FDA Triggers: SaMD, CDS, And AI Lifecycle Considerations
Some healthcare software is regulated as a medical device, and some is not. The line matters.
The FDA describes Software as a Medical Device (SaMD) as software intended to be used for one or more medical purposes that performs those purposes without being part of a hardware medical device.
If your product includes clinical decision support, the FDA also publishes guidance that clarifies the scope of oversight for certain CDS software functions.
If AI is part of your roadmap, note that the FDA has been active on AI-enabled device software lifecycle topics, including draft guidance focused on lifecycle management and marketing submission recommendations (January 2025).
Buyer move: do a lightweight regulatory classification exercise in discovery. It can save months later.
When GDPR Or UK GDPR Applies
If you serve users in Europe or the UK, health data is considered sensitive and often requires additional conditions beyond a standard lawful basis.
Buyer implication: data minimization, consent flows, retention controls, and data subject rights handling may become first-class scope items.
SOC 2 As A Procurement Expectation
Even when SOC 2 is not legally required, it frequently appears in procurement and security questionnaires.
If you are selling to providers, payers, or enterprise clients, expect questions about controls relevant to security, availability, processing integrity, confidentiality, or privacy.
If you are selling to providers, payers, or enterprise health tech, plan for SOC 2 readiness, or at least a clear security control story aligned to it.
Compliance Mapping Table
| Requirement Area | What Buyers Expect In The Product | What They Expect In Delivery |
|---|---|---|
| HIPAA Safeguards | RBAC, audit logs, encryption, secure auth | Risk analysis support, security testing |
| Breach Readiness | Monitoring, logs, access traceability | Incident response plan, clear ownership |
| Interoperability | FHIR-based APIs, data export patterns | Integration proofs, data mapping |
| FDA Risk | Guardrails for decision-making features | Classification review, documentation plan |
| GDPR Or UK GDPR | Consent, minimization, retention controls | DPIA support, privacy-by-design workflows |
| SOC 2 Expectations | Strong security posture and controls | Evidence, policies, repeatable processes |
Cost: How To Budget For Custom Healthcare Software Development Services
Cost questions are normal. The mistake is expecting one number before you have enough inputs.
Instead, budget in layers: MVP now, compliance-ready foundations, then phased expansion.
The Cost Buckets That Matter
Typical Cost Ranges (2025–2026 Benchmarks)
When you ask for an estimate, make sure it includes (or clearly excludes) these buckets:
- Discovery and requirements (workflows, data boundaries, integration plan)
- UX and product design (especially for clinician usability)
- Core engineering (web, mobile, backend, APIs)
- Integrations (EHR, labs, payers, devices, SSO)
- Security engineering (threat modeling, hardening, penetration testing strategy)
- Compliance documentation and audit readiness activities
- QA and validation (including edge cases, role testing, audit trails)
- DevOps and infrastructure (environments, CI/CD, monitoring)
- Post-launch stabilization and maintenance
SeeSaw Labs positions custom software development around scalable, secure architecture and includes HIPAA and SOC 2 compliance capability as part of its delivery scope, which is exactly the kind of capability buyers should look for.
Ballpark Ranges (And What They Assume)
Here are market ranges you will commonly see referenced, but treat them as starting points, not commitments:
- MVPs are often quoted in the mid-five-figures to low six-figures range. One 2025 guide cites typical MVP budgets of around $70K to $120K and timelines of 4 to 6 months.
- HIPAA-oriented builds can range widely. One HIPAA development guide cites $30,000 to $400,000+, depending on functionality and integrations.
- A custom software cost benchmark article provides an example range for healthcare software of $75,000 to $250,000+, depending on HIPAA compliance, integration, and security needs.
What these ranges usually assume:
- You are building a defined MVP, not a full EHR replacement
- Integration scope is controlled and staged
- Compliance work is planned, not bolted on late
Pricing Models: Fixed Scope Vs Time And Materials
Most healthcare buyers end up choosing one of these:
- Fixed scope, fixed price: best when requirements are stable, and risk is low
- Time and materials: best when learning is part of the plan (common in healthcare MVPs)
- Dedicated team: best when you have a clear product roadmap and want continuity
A good partner will help you choose a model based on uncertainty, not just preference.
Hidden Costs And How To Avoid Surprise Spend
Common “budget surprises” in healthcare include:
- EHR integration complexity, including sandbox delays, mapping, and credentialing
- Data migration and normalization
- Security and compliance rework when discovered late
- Clinical workflow iteration, because what works in a demo may fail in a busy clinic
- Adoption support and training materials
A simple way to reduce surprise spending is to properly fund discovery. Even a short planning phase can prevent months of rework.
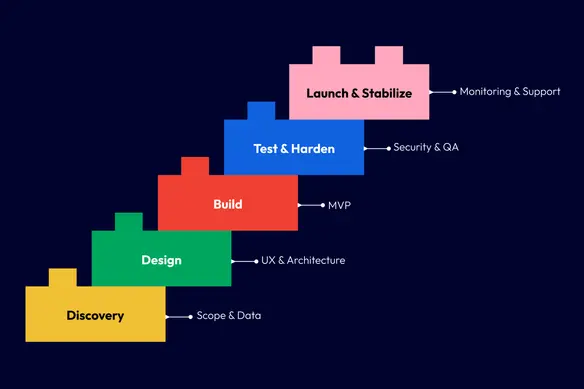
Timeline: A Realistic Delivery Plan From Discovery To Launch
Healthcare timelines slip when teams underestimate integration, compliance evidence, and real-world workflow iteration.
Typical Phase Breakdown
- Discovery and definition (2 to 6 weeks)
Includes workflow mapping, data boundaries, integration plan, and compliance triggers. - Design (2 to 6 weeks)
User flows, prototypes, clinical usability review, and early architecture decisions - MVP build (8 to 16+ weeks)
SeeSaw Labs notes many MVPs launch within 12 to 16 weeks, depending on scope. - Testing and security hardening (2 to 6 weeks, overlaps with build)
- Launch and stabilization (2 to 4 weeks)
- Roadmap iterations (ongoing)
A separate industry guide notes that typical healthcare MVP builds can fall within a 4- to 6-month window, especially as features and compliance needs grow.
Timeline Table
| Phase | Typical Duration | Key Deliverables |
|---|---|---|
| Discovery | 2–6 Weeks | Requirements, data map, compliance triggers, MVP scope |
| Design | 2–6 Weeks | UX flows, clickable prototype, architecture outline |
| Build | 8–16+ Weeks | Working product increments, integrations in stages |
| Test And Harden | 2–6 Weeks | QA, security testing, audit logging validation |
| Launch | 1–2 Weeks | Production release, monitoring, and rollback plan |
| Stabilize | 2–4 Weeks | Bug fixes, performance tuning, adoption feedback |
What Extends Timelines In Healthcare
Plan extra time if you have:
- EHR integrations or complex interoperability requirements (FHIR mapping, multiple systems)
- Multi-role permissioning and audit trails (providers, staff, patients, admins)
- FDA risk classification considerations (SaMD, CDS behavior)
- International privacy obligations (GDPR and UK GDPR)
- Enterprise procurement requirements (SOC 2, security questionnaires)
If you are a startup, SeeSaw Labs’ launch-focused guide to launching a healthcare product is a helpful resource you can follow.
Choosing The Right Development Partner
In healthcare, the best partner is the one that prevents expensive mistakes, not just the one that can code.
SeeSaw Labs describes a 5D process (Discover, Define, Design, Develop, Delight), which is a useful mental model for what a strong partner should operationalize, especially the early stages that de-risk scope and compliance.
A Healthcare Vendor Evaluation Checklist
Use this checklist when you compare vendors:
- Healthcare experience: Can they explain real workflows, not just features?
- Compliance approach: Can they map HIPAA safeguards to product controls and delivery steps?
- Security engineering: Do they talk about threat modeling, logging, and incident response readiness?
- Interoperability: Can they discuss FHIR and how they validate integrations?
- FDA awareness: Can they spot when you might drift into SaMD or regulated CDS?
- Quality and testing: Do they have a clear test strategy for roles, workflows, and auditability?
- Ownership and handoff: Will you own code, infrastructure, and documentation?
- Post-launch support: do they plan for monitoring, updates, and ongoing optimization?
Questions To Ask In Calls And RFPs
Ask these and listen for specific answers:
- Where does compliance show up in your delivery process, and what artifacts do you produce?
- How do you handle PHI boundaries, least-privilege access, and audit logging?
- What is your approach to EHR integrations and sandbox validation?
- What does security testing look like before launch?
- What is included in your estimate, and what commonly gets missed?
- How do you handle scope changes without breaking trust?
Frequently Asked Questions
What Are Custom Healthcare Software Development Services?
They are services that design, build, integrate, and maintain software tailored to healthcare workflows, often including security, compliance planning, and interoperability needs.
How Do I Know If My App Needs To Be HIPAA Compliant?
If your product creates, receives, maintains, or transmits ePHI for US covered entities or business associates, HIPAA Security Rule safeguards become central.
Do I Need HL7 FHIR For My Project?
Not always. You typically need FHIR when integrating with modern EHR APIs or when exchanging standardized healthcare data.
How Long Does It Take To Build A Healthcare MVP?
It depends on the scope and integrations. Some teams cite 4 to 6 months for a typical healthcare MVP, while others can launch in 12 to 16 weeks with tight scope control.
How Much Does Custom Healthcare Software Usually Cost?
It varies widely based on features, integrations, and compliance needs. Published estimates range from tens of thousands to several hundred thousand dollars for HIPAA-oriented builds, depending on complexity.
When Does The FDA Get Involved With Healthcare Software?
When software is intended for medical purposes that may qualify it as SaMD, or when regulated CDS behaviors are involved. It is worth assessing early, even if you do not plan to submit immediately.
Conclusion
Custom healthcare software projects go smoothly when you treat scope, compliance, cost, and timeline as a single, interconnected system. Define what you are building, identify the regulations and standards that apply, and choose a delivery plan that produces evidence, not just features.
Key Takeaways
- Scope drives everything, especially data boundaries and integrations.
- Compliance must be designed into the product and the delivery process, not added at the end.
- Budget and timeline become predictable when you fund discovery, stage integrations, and commit to an MVP-first roadmap.